RAISING THE BAR
IN PHARMACEUTICALS

Because When Every Dose Matters, Only the Best Will Do.
Across the country, healthcare facilities are facing critical drug shortages that disrupt treatment and put patient safety at risk.
At Turbare, we’re closing the gap with reliable access to high-quality compounded medications produced under the strictest standards.
As an FDA-registered 503B outsourcing facility, Turbare is built for precision and scale. From sterile IV medications to shortage-response preparations, we operate under full current good manufacturing practices (cGMP) to ensure that every dose we deliver meets the highest bar for quality, sterility, and compliance.
Why Clinicians Choose Turbare
FDA-Registered 503B Outsourcing Facility
Our status allows your hospital to meet all compliance and patient safety requirements with confidence.
Current Good Manufacturing Practices (cGMP) Environment
We ensure that every preparation meets the highest federal and industry standards.
Fully Automated Production Systems
Count on accuracy and reproducibility in every sterile batch.
Responsive Drug-Shortage Support
We give your clinicians access to essential compounded drugs when others can’t supply them.
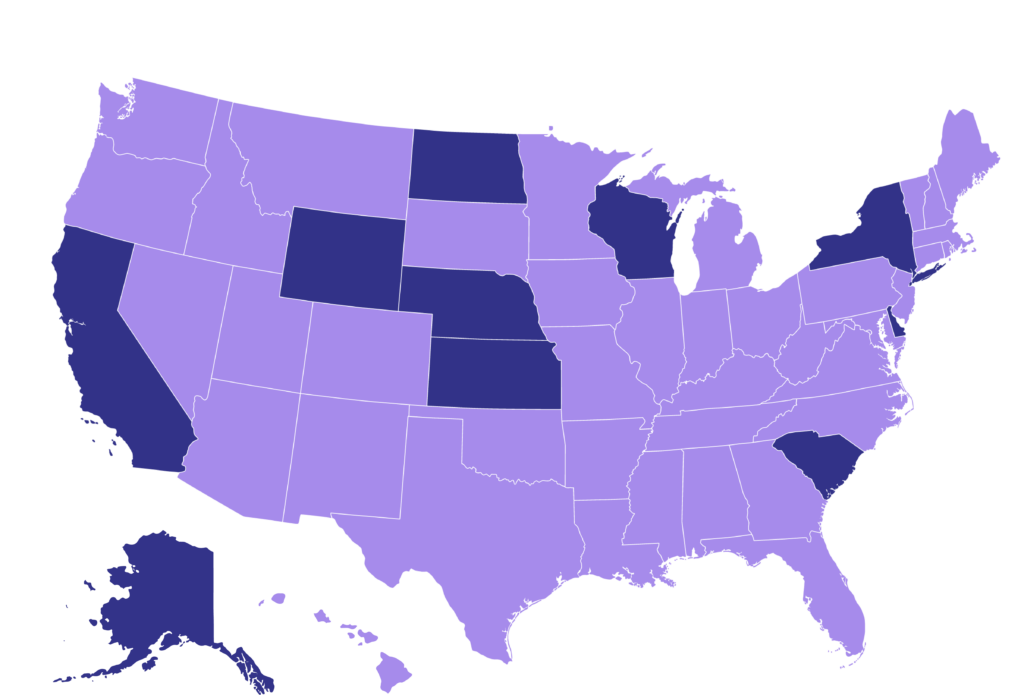
Nationwide Distribution and Expanding Partnerships
Healthcare facilities of all sizes can depend on a consistent sterile medication supply.
Expert Team of Pharmacists and Quality Specialists
Get guidance from professionals who understand compliance and care.
Sterile Compounding for Performance & Accountability
Turbare provides compounded drugs and sterile drugs to hospitals, clinics, and healthcare facilities nationwide. Our 44,000+ sq. ft. automated facility in Conway, Arkansas, is designed for performance and accountability—combining advanced cleanroom environments with rigorous testing to protect the integrity of every product.
Unlike a traditional compounding pharmacy, Turbare functions as a federally regulated outsourcing facility under the Federal Food, Drug, and Cosmetic Act and Federal Drug Quality and Security Act, ensuring traceability, documentation, and consistency that meet the most demanding healthcare standards.

Our Promise
At Turbare, we hold ourselves to one standard: Only the best will do.
Every process, from formulation to final packaging, follows strict current good manufacturing practices and exceeds the standards applied to other compounding pharmacies.
Our team of pharmacists, engineers, and quality professionals ensures that every vial, syringe, and IV unit we produce is safe, sterile, and consistent—because every patient matters. We maintain full alignment with federal drug quality requirements and the Federal Food, Drug, and Cosmetic Act, giving our partners peace of mind that they’re working with one of the nation’s most trusted 503B outsourcing facilities.
When healthcare systems choose Turbare, they’re choosing reliability, transparency, and excellence in every compounded solution.
Partner with Our Experts
Join healthcare leaders nationwide who trust Turbare to deliver sterile compounded medications that uphold the highest standards of quality and patient safety.
Contact our team to discuss your needs, product availability, and how our outsourcing facility can strengthen your supply chain.
Where Can We Ship?